The common disease of Dairy cows and treatments
Uterine Disease in Dairy Cows
Those cows with problems during the peripartum period exhibit a reduced ability to control uterine infections (Noakes et al., 2002). In general, any condition which affects postpartum immunity, such as for example the administering of progesterone or glucocorticoids, is transformed into an underlying factor (Sheldon and Dobson, 2004). Among other risk factors established for these infections include the level of hygiene of the environment, birth (particularly in the case of twins), caesarean, retention of the placenta and traumatism in the genital tissue during dystocia or manipulation (Bell and Roberts, 2007). Some metabolic conditions such as milk fever, ketosis and displacement of the abomasum to the left have also been associated with these problems (Sheldon and Dobson, 2004).
The most common postpartum uterine diseases are: endometritis, metritis and pyometra. The diagnostic criteria vary according to researchers, and in spite of the fact that efforts have been made to standardize the concepts, these have been unsuccessful (Leblanc et al., 2002; Sheldon et al., 2006; Azawi, 2008).
Metritis
Metritis is an inflammatory process which affects all layers of the lining of the uterus: endometrium, submucosa, muscular and serous membrane (BonDurant, 1999). According to clinical signs, it may be classified as puerperal metritis and clinical metritis (Sheldon et al., 2006). A case of puerperal metritis is defined as a cow with an abnormally enlarged uterus and a uterine discharge which is watery, fetid and reddish-brown in color, associated with signs of systemic disease such as decreased production, depression and fever, within 21 days after giving birth.

Cloudy and bloody vulvar discharge
For its part, in the case of clinical metritis this is defined as a cow which exhibits no signs of systemic disease, but which does have the uterus abnormally enlarged and a purulent uterine discharge detectable in the vagina within 21 days after giving birth (Lewis, 1997; Sheldon et al., 2006).
Puerperal metritis is the only uterine infection capable of threatening the life of the animal (toxic-septic puerperal metritis) (Lewis, 1997; Melendez et al., 2004) and very often systemic treatments are required because the endotoxins and pathogens can leave the uterus and enter the circulation when the mucous membrane is severely weakened. Frequently, this occurs together with vaginitis and cervicitis, and if this infection manages to spread through the wall of the uterus it can cause perimetritis and peritonitis (Christensen et al., 2009).
Endometritis
This is an inflammation of the surface of the endometrium which does not extend beyond the spongy strata and the underlying glandular tissue, with histological evidence of inflammation (Sheldon et al., 2006). This process is characterized by degenerative changes in the superficial epithelium, vascular congestion with edema in the stroma and migration of neutrophils and other inflammatory cells to the affected area (Foldi et al., 2006).
During the puerperal period 90% of cows develop moderate endometritis. In the majority of cows the local defense mechanisms manage to eliminate the infection and the problem is resolved in a few days, but when the infection persists for more than 21 days (Sheldon et al., 2004), for example, as a consequence of acute metritis, and the infection continues, a case of clinical endometritis may result. The most common pathogens in this type of case are: Arcanobacterium pyogenes and anaerobic gram-negative bacteria (Fusobacterium necrophorum, Prevotella and Bacteroides ssp.). (Foldi et al., 2006)
Clinically, endometritis is characterized by the presence of purulent or mucupurulent discharge in the vagina 21 days or more after giving birth. It is frequently associated with uterine involution retardation and is not accompanied by systemic clinical signs (Sheldon et al., 2006).
The diagnostic criteria for endometritis in the postpartum period of dairy cows have been validated examining the factors associated with an increase in the birth-to-conception interval (Leblanc et al., 2002). Among the most significant signs are: the presence of a purulent vaginal discharge or a cervical diameter greater than 7.5 centimeters and the presence of mucopurulent content in the vagina, 21 days or more after giving birth (Sheldon and Noakes, 1998; Sheldon et al., 2006). In spite of the fact that the clinical signs lead us to the diagnosis of endometritis, the definitive diagnosis is made based on the histological evaluation of endometrial biopsies. In field conditions, the direct examination of the vagina with a speculum and rectal palpation are the most commonly-used techniques (Sheldon et al., 2006).
It should be stressed that the diagnosis of an animal with clinical endometritis less than 21 days after giving birth could include animals which are capable of spontaneously resolving the bacterial contamination. Also, the normal elimination of lochia may complicate diagnosis during this time period (Le Blanc et al., 2002).
On the other hand, subclinical endometritis, also known as chronic, is an endometrial inflammatory process that affects the reproductive and productive performance of cows, and is usually diagnosed through cytology given the absence of purulent material in the vagina (Sheldon et al., 2006; Foldi et al., 2006).
The cytological diagnosis of endometritis is based on the proportion of neutrophils found when the uterine lumen is washed using a cytobrush. A case of subclinical endometritis is defined as the discovery of a quantity of neutrophils greater than 18% in cytological samples collected from the uterus between 21 and 33 days after giving birth or a proportion of neutrophils greater than 10% in samples collected between 34 and 47 days after giving birth, in the absence of clinical endometritis. (Gilbert et al., 2005, Sheldon et al., 2006).
The main consequence of endometritis is not only infertility during the period of infection, but also the reduced fertility produced even after the successful resolution of the disease. In studies of cows treated successfully for endometritis, the conception rate is 20% lower compared with healthy animals, and in addition 3% of the total number of cows remains infertile. This situation brings with it as a direct consequence considerable financial losses for the producer (Herath et al., 2006).
Pyometra
Pyometra is a process characterized by the accumulation of purulent or mucupurulent material within the uterine lumen with the presence of an active corpus luteum. Due to the presence of luteal progesterone the cervix is closed (Sheldon et al., 2006), although in some cases the lumen is not completely occluded and a purulent discharge can be observed in the vagina (Sheldon et al., 2004). In the case of pyometra, through rectal palpation the uterine horns are found to be enlarged as a result of their content. It is important to diagnose pregnancy before applying treatment.
In most cases, pyometra occurs as a consequence of endometritis, because when cows with this problem ovulate, they develop pyometra, and for this reason it is detected almost exclusively in cows with an active corpus luteum, from 21 days after giving birth (Foldi et al., 2006).
Early ovulation after giving birth may predispose the cow to the emergence of this problem (Sheldon et al., 2006). If the birth-first ovulation interval is very short this could lead to pyometra, because the colonies of A. pyogenes and anaerobic gram-negative bacteria remain within the uterus after ovulation, enabling continued bacterial growth after the formation of the corpus luteum (Sheldon et al., 2004).
Pyometra may also be produced when:
- Fetal death occurs, followed by the invasion of bacterial pathogens such as A. pyogenes and the persistence of the corpus luteum of gestation (Sheldon et al., 2004).
- An infection is produced during handling in the case of artificial insemination at incorrect time (Sheldon et al., 2004).
Animals affected do not look sick, but when in anestrus they may be presented to the veterinarian for diagnosis of pregnancy and it is in this moment when the problem is detected. After resolution, the cows take time to become pregnant, and this is directly related to the endometrial degeneration which increases the rate of embryonic and fetal losses (Sheldon et al., 2004).
Treatment
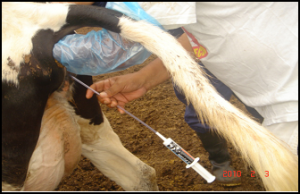
The traditional treatments of endometritis and metritis are based on the infusion of antimicrobials in the uterine cavity. There has been much controversy regarding the effectiveness of this type of protocol, given that antibiotics can reach the circulation and leave residues in the milk (Lewis, 1997). However, in a study made by Alvarado et al (2010) it was concluded that the residue in the milk resulting from an intra-uterine antibiotic infusion based on Cefalexin, Neomycin, Cloxacillin and Vitamin A (Metri-Cef 3®) in dairy cows during lactation is nil; therefore, the results of the Delvotest SP method were negative.

Metri – Cef 3 syringe and infusion tube
The systemic application of antibiotics is important in severe cases of septic metritis, given that the infection may become generalized (Sheldon et al., 2004). When puerperal metritis develops, the application of active antibiotics against A. pyogenes, E. coli and anaerobic gram-negative bacteria is necessary for the achieving of successful treatment against massive uterine infections and to prevent septic complications (Melendez et al., 2004).
The rational treatment of endometritis has been widely discussed (Azawi, 2008) and is aimed at improving fertility through the elimination of the pathogens in the uterus. The most commonly-used treatments number three: PGF2a or similar when there is corpus luteum present, intra-uterine antibiotics and estrogens (Sheldon et al., 2004). Treatment with PGF2a is as effective as other treatments, as well as being less aggressive and the milk need not be discarded (Lewis 1997, Sheldon and Dobson, 2004). In the absence of corpus luteum, a broad range of intra-uterine treatments have been administered, including antiseptics and antibiotics.
The administering of estradiol at a dose of 5-10 milligrams per animal has been used therapeutically for postpartum endometritis and is as effective as PGF2a (Sheldon and Noakes, 1998), and both are superior to the spontaneous recovery of untreated animals. However, the treatment-conception interval is much longer with estradiol than with PGF2a or the administering of intra-uterine antibiotics (Sheldon and Noakes, 1998). In spite of the potential of estrogens in the treatment of endometritis, the results have been incorrect and vary according to the pharmacological presentation and the dose administered. Also, the authorities of several countries have banned the use of estrogens (Sheldon et al., 2004).
The treatment of pyrometra is universal and researchers agree that PGF2a or similar is the most appropriate treatment, given that it produces luteolysis, and the presence of the corpus luteum is one of the most significant underlying factors of this condition (Sheldon et al., 2004).
Conclusions
- In the uterus of all cows postpartum bacterial contamination occurs, but this does not necessarily lead to infection or the development of uterine disease.
- The main uterine diseases of the postpartum period are: metritis, endometritis and pyometra.
- The main consequences of endometritis are infertility and reduced infertility, even after the successful resolution of the disease.
- The treatments of metritis and endometritis include the application of antibiotics, including both parenteral and intra-uterine application. In cases of endometritis PGF2a has also been recommended.
- The treatment of pyometra is universal and consists of the application of PGF2a.
References:
- Alvarado A, Tang J, Ruiz F, Rodriguez L. 2010. Evaluación de la Residualidad en Leche de una Infusión Antibiótica Intrauterina sobre la base de Cefalexina, Neomicina, Cloxacilina y Vitamina A (Metricef 3®) en vacas lecheras del distrito de Pachacamac, departamento de Lima. [Internet], [10 Noviembre 2011]. Available in: [http://www.agrovetmarket.com/pdf/antibiotico/Metricef/Trabajo%20de%20Investigaci%F3n/Metricef%203%20residualidad%20leche.pdf.
- Azawi OI. 2008. Postpartum uterine infection in cattle. Animal Reproduction Science 105: 187-208.
- Bell MJ, Roberts DJ.2007. The impact of uterine infection on a dairy cow’s performance. Theriogenology 68: 1074-1079.
- Bondurant R. 1999. Inflammation in the bovine female reproductive tract. J. Anim. Sci. 77, 101-110.
- Christensen BW, Drost M, Troedsson MHT. 2009. Disease of the Reproductive System En: Smith BP, ed. Large animal internal medicine. 4th ed. USA. Mosby-Elsevier. p 1419-1483.
- Földi J, Kulcsár M, Pécsi A, Huyghe B, de Sa C, Lohuis JACM, Cox P, Huszenicza G. 2006. Bacterial complications of postpartum uterine involution in cattle. Animal Reproduction Science 96: 265-281.
- Gilbert RO, Shin ST, Guard CL, Erb HN, Frajblat M.2005. Prevalence of endometritis and its effects on reproductive performance of dairy cows. Theriogenology 64: 1879-1888.
- Herath S, Dobson H, Bryant CE, Sheldon IM.2006. Use of the cow as a large animal model of uterine infection and immunity. Journal of Reproductive Immunology 69: 13-22
- LeBlanc SJ, Duffield TF, Leslie KE, Bateman KG, Keefe GP, Walton JS, Johnson WH.2002. Defining and diagnosing postpartum clinical endometritis and its impact on reproductive performance in dairy cows. J Dairy Sci 85: 2223-2236.
- Melendez P, McHale J, Bartolome J, Archbald LF, Donovan GA.2004. Uterine involution and fertility of Holstein cows subsequent to early postpartum PGF2a treatment for acute puerperal metritis. J Dairy Sci 87: 3238-3246.
- Lewis GS. 1997. Uterine health and disorders. J Dairy Sci 80: 984-994.
- Noakes DE, Parkinson TJ, England GCW, Arthur GH. 2002. Arthur’s Veterinary Reproduction and Obstetrics, eighth ed. Elsevier Sci. Ltd, 868 p
- Sheldon y Noakes. 1998. Comparison of three treatments for bovine endometritis. Veterinary Record 142: 575-579.
- Sheldon IM, Dobson H.2004. Postpartum uterine health in cattle. Animal Reproduction Science 82-83: 295-306.
- Sheldon IM, Barret DC, Boyd H.2004. The postpartum period. En: Andrews A, Blowey R, Boyd H, Eddy R, eds. Bovine Medicine Diseases and Husbandry of Cattle. USA. Blackwell Science Ltd. p 508-538.